Products
Dedicated to building a globally leading R&D and manufacturing platform for the deep processing of bismuth materials.
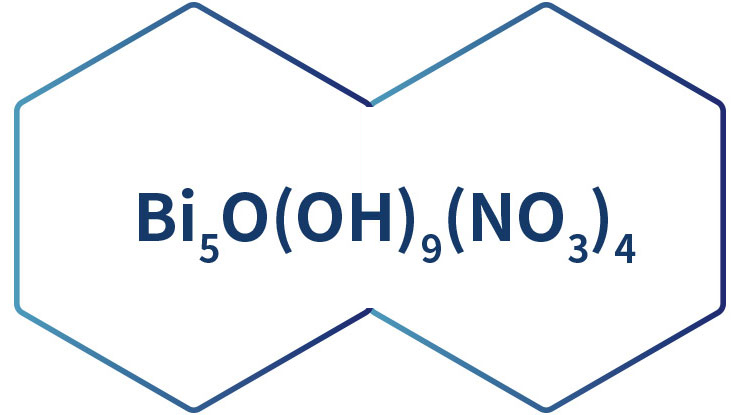
Hexagonal crystalline flakes or microcrystalline powder. Odorless. Tasteless.Density 4.928g/cm³. Soluble in dilute hydrochloric acid and nitric acid; insoluble in water and ethanol.
In the pharmaceutical field, it is used as an active pharmaceuti cal ingredient (API) for the preparation of bismuth-based drugs, commonly used to treat gastritis, diarrhea, and other gastroin testinal diseases. It can also be used to prepare veterinary medicines for treating diarrhea in animals. In the industrial field, it can be used as a catalyst for electrophoretic coatings.
| Bismuth Subnitrate(USP) | Bismuth Subnitrate(EUROPEAN PH.) | ||
|---|---|---|---|
| Determinations | Specifications | Determinations | Specifications |
| Appearance | White powder | Appearance | White or off-white powder |
| Identification | Bismuth Nitrate | Identification | A,B: Bismuth |
| Oxide Bismuth Content ODB | ≥79.0% | C:Nitrate | |
| Loss on Drying | ≤3.0% | D:pH | |
| Carbonate | Test | Acidity | Test |
| Chloride | ≤350ppm | Chlorid | ≤200ppm |
| Sulphate | Test | Pb | ≤20ppm |
| Cu | Test | Cu | ≤50ppm |
| Ag | Test | Ag | ≤25ppm |
| Pb | Test | Non precipitated Substances in ammonia | ≤1.0% |
| As | ≤8ppm | Loss on Drying | ≤3.0% |
| Ammonium Salts | Test | Bismuth Content(ODB) | 71.0-74.0% |
| Alkaline Earth | ≤0.5% | ||